G2(+)M Study on N-Alkylamino Cation Affinities of Neutral Main-Group Element Hydrides: Trends Across the Periodic Table
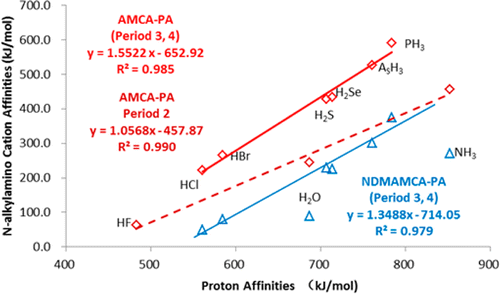
We have made an extensive theoretical exploration of gas-phase N-alkylamino cation affinities (NAAMCA), including amino cation affinities (AMCA) and N-dimethylamino cation affinities (NDMAMCA), of neutral main-group element hydrides of groups 15–17 and periods 2–4 in the periodic table by using the G2(+)M method. Some similarities and differences are found between NAAMCA and the corresponding alkyl cation affinities (ACA) of HnX. Our calculations show that the AMCA and NDMAMCA are systematically lower than the corresponding proton affinities (PA) for HnX. In general, there is no linear correlation between NAAMCA and PA of HnX. Instead, the correlations exist only within the central elements X in period 2, or periods 3–4, which is significantly different from the reasonable correlations between ACA and PA for all HnX. NAAMCA (HnX) are weaker than NAAMCA (Hn–1X–) by more than 700 kJ/mol and generally stronger than ACA (HnX), with three exceptions: H2ONR2+(R = H, Me) and HFNH2+. These new findings can be rationalized by the negative hyperconjugation and Pauli repulsion.
