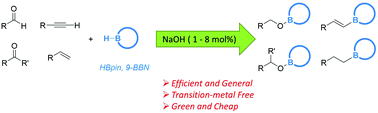
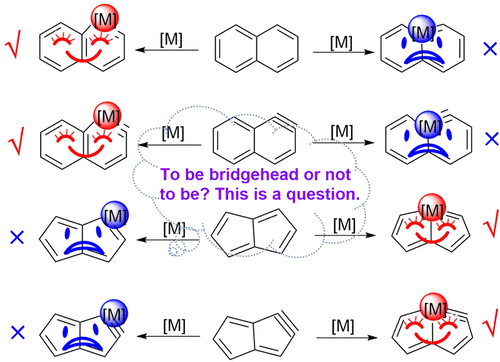
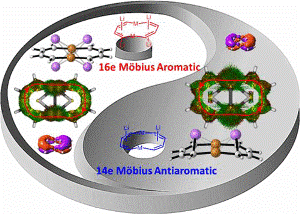
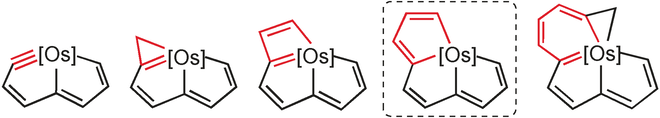
Direct energetic evaluation of aromaticity by cleaving the rings of cyclic compounds
Submitted by Jun Zhu on Sat, 09/01/2018 - 10:33
Aromaticity, one of the central topics in chemistry, has attracted continuing interest of both experimentalists and theoreticians.