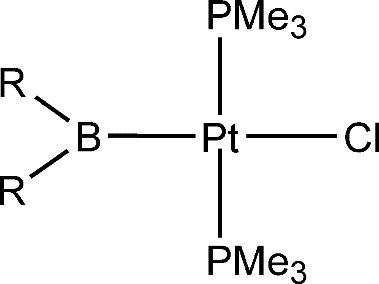
Insertion Reactions of Allenes Giving Vinyl Complexes
Submitted by Jun Zhu on Thu, 10/31/2013 - 23:35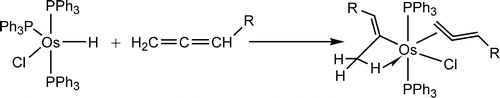
Treatment of OsHCl(PPh3)(3) with allenes CH2=C=CHR at room temperature in benzene produced the vinyl complexes OsCl(C(CH3)=CHR)(CH2=C=CHR)-(PPh3)(2), instead of eta(3)-allyl complexes as normally observed. DFT calculations show that the formation of the vinyl complex is favored kinetically.