Direct Conversion of N2 and O2 to Nitric Oxide at Room Temperature Initiated by Double Aromaticity in the Y2BO+ Cation
Submitted by Jun Zhu on Mon, 11/14/2022 - 16:56
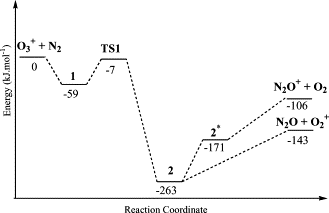
The conversion of dinitrogen to more useful and reactive molecules has been the focus of intense research by chemists. In contrast to reductive N2 fixation, direct oxidation of N2 by O2 to nitric oxide under mild conditions via a thermochemical process is extremely challenging. Herein, we report the first example of N2 and O2 activation and coupling under thermochemical conditions through the remarkable ability of Y2BO+ to react with one N2 and two O2 molecules.