Nucleophilic reactions of osmanaphthalynes with PMe3 and H2O
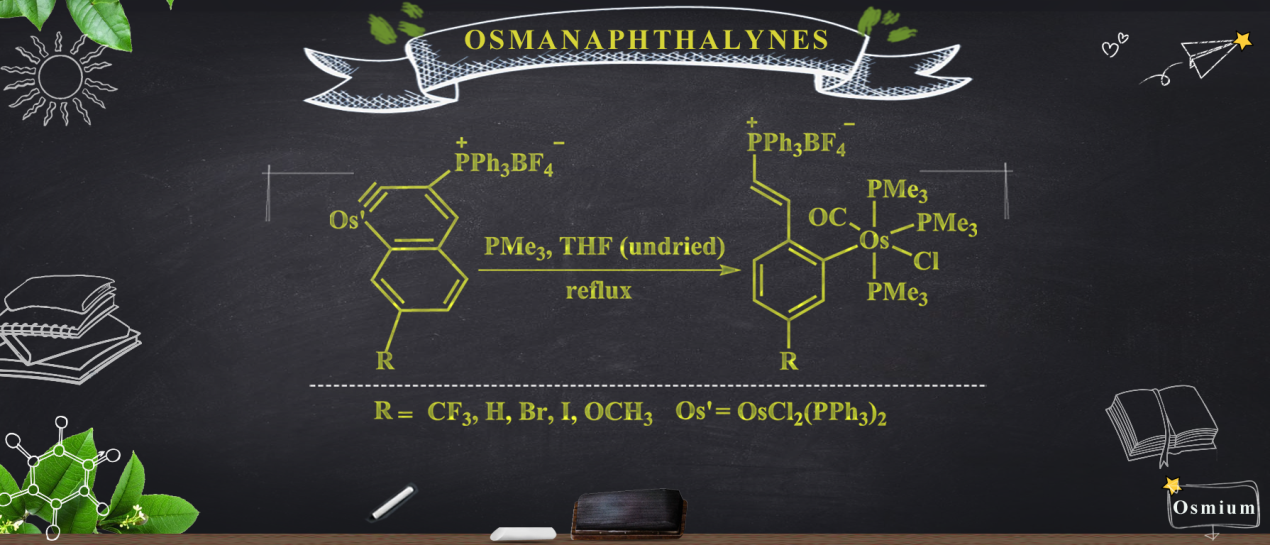
Members of a new class of complexes, 2(CF3), 2(H), 2(Br), 2(I), and 2(OCH3), have been synthesized by a one-pot method involving the treatment of osmanaphthalynes bearing corresponding substituents (1(CF3), 1(H), 1(Br), 1(I), and 1(OCH3) with trimethylphosphine (PMe3) and water (H2O). The main reaction process involves two steps, namely a ligand-exchange reaction with trimethylphosphine and nucleophilic addition of water to the Os≡C bond of the osmanaphthalyne. The substituents have a significant influence on the rate of the reaction, as befits a nucleophilic addition. Fortunately, the key intermediate [1(OCH3)]′ could be successfully captured, and the detailed reaction mechanism has been explored with the aid of density functional theory (DFT) calculations, in excellent agreement with the experimental findings. All of the target complexes have been fully characterized by 1H, 31P{1H}, and 13C{1H} NMR spectroscopies, high-resolution mass spectrometry, and elemental analysis.
https://chemistry-europe.onlinelibrary.wiley.com/doi/10.1002/chem.202100176
