Strategy for the Synthesis of Inverse Sandwich 1,4-Diarylbutadiene Dianionic Rare-Earth Metal Complexes via Directing Coupling of β-Aryl Vinyl Boron Reagents
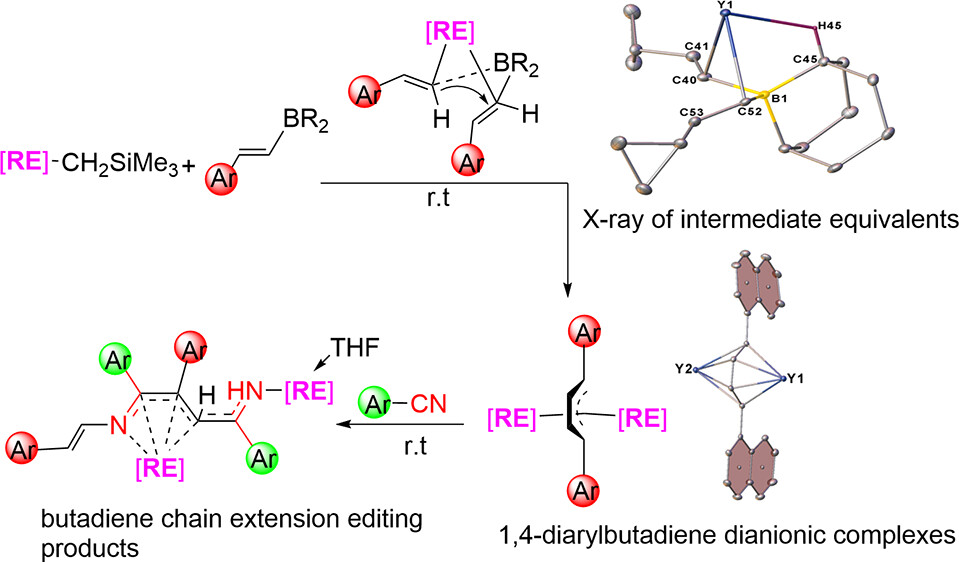
Aryl and vinyl boron reagents are widely employed in modern organic synthesis. However, the direct coupling of nucleophilic vinyl boron reagents to construct conjugated 1,4-diarylbutadiene dianionic (DABDA) motifs in the absence of additives remains a significant challenge. Here, we report for the first time the direct coupling of various β-aryl vinyl boron reagents, initiated by the rare-earth metal monoalkyl complexes for the synthesis of a number of rare-earth metal complexes featuring inverse sandwich DABDA motifs. Isolation of key intermediates (or intermediate equivalents) and theoretical results provide mechanistic insights involving deboronative transmetalation, followed by 1,2-metalate shift of the vinyl group of the metal-bonded tetracoordinate borates, and deboronation. DFT calculations indicate that the singlet state, with two electrons in the butadiene π* orbital, is more stable than the triplet state, where the electrons are localized in the π* orbitals of the aryl and butadiene parts. The variable-temperature magnetic susceptibility further confirmed this result. The DABDA complexes exhibit unprecedented redox reactivity in their reactions with unsaturated compounds including bipyridine, azobenzene, trimethylsilyl azide, imines, ketones, and nitriles. Notably, a butadiene chain extension editing process was also unveiled upon treatment of the DABDA complexes with aromatic nitriles.
