Pd-catalyzed highly regio-, diastereo-, and enantioselective allylic alkylation of α-fluorophosphonates
Submitted by Jun Zhu on Tue, 06/03/2014 - 13:07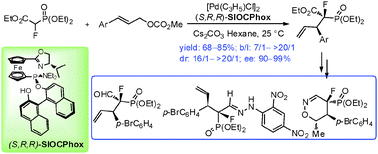
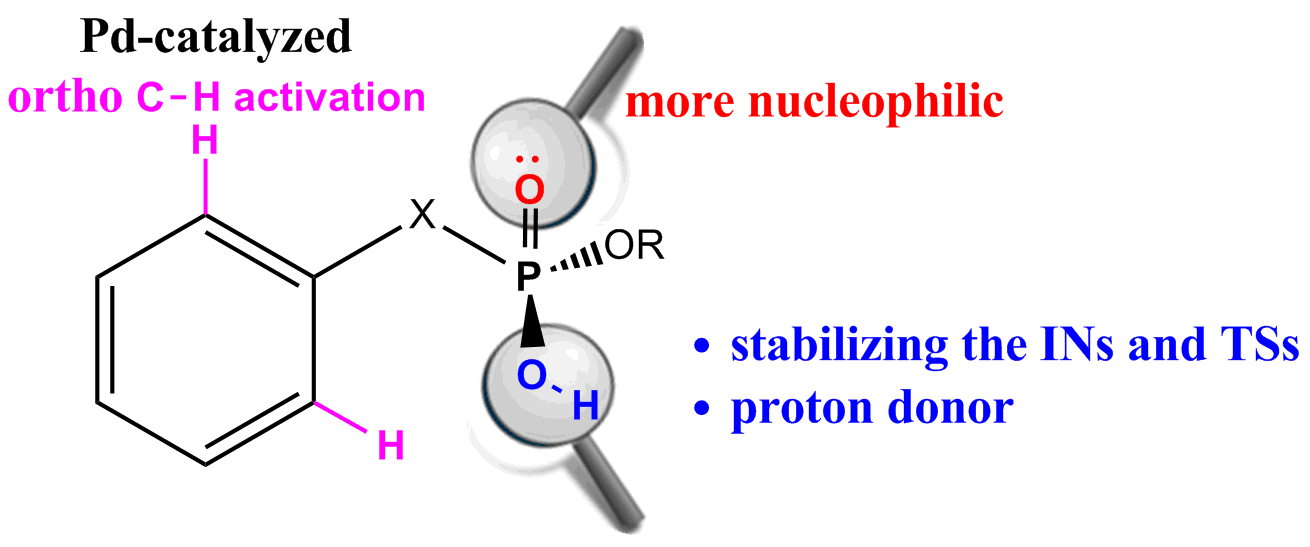
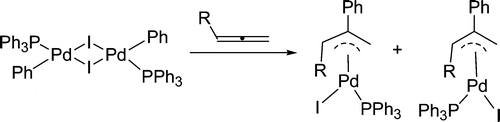
Highly efficient Pd-catalyzed asymmetric allylic alkylation reaction of ethyl-2-fluoro-2-(diethoxyphosphoryl)acetate with monosubstituted allylic substrates has been developed, affording corresponding α-fluorophosphonates with two chiral centers in high regio-, diastereo- and enantio-selectivities. The usefulness of the products in organic synthesis has been demonstrated.
http://pubs.rsc.org/en/content/articlelanding/2014/cc/c4cc02158d#!divAbstract