Probing a General Rule towards Thermodynamic Stabilities of Mono BN-doped Lower Polyenes
Submitted by Jun Zhu on Thu, 02/09/2017 - 15:55
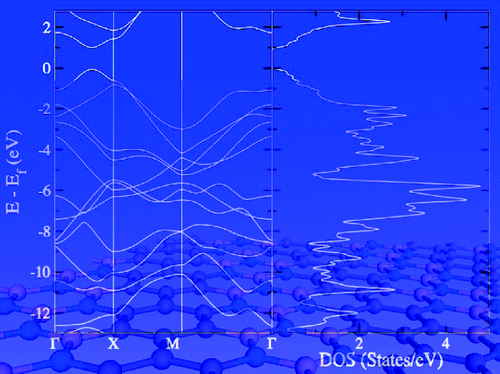
The BN-doped organic analogues are interesting as aliphatic amineboranes for hydrogen storage, precursors for aromatic borazines and adsorbent cage azaboranes. However, BN-doped aliphatic polyenes remained undeveloped. Herein, we perform theoretical calculations on two mono BN-doped aliphatic lower polyenes, 1,3-butadiene and 1,3,5-hexatriene. A general rule is proposed, i.e., isomers with terminal nitrogen and directly BN-connected, N−B(R), in particular, are of significant thermodynamic stability as compared with their inverse analogues (where boron is at the terminal position).