Exploiting heterocycle aromaticity to fabricate new hot exciton materials
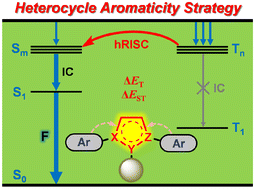
Efficiently harvesting electroluminescent triplet excitons is of great importance for practical applications of organic light-emitting diodes (OLEDs). Hot exciton materials are regarded as the up-and-coming new generation luminogens and hold unique advantages for achieving high efficiency electroluminescent devices. Therefore, exploring a new molecular design strategy for developing new hot exciton materials remains a challenging yet fascinating task so far. In the current contribution, we present a novel design strategy for exploiting heterocycle aromaticity to fabricate new hot exciton materials from both theoretical and experimental perspectives. By virtue of heterocycle aromaticity and peripheral functionalization, molecular excited state energetics could be rationally modulated to achieve the desired energy gaps of singlet–triplet and triplet–triplet states and consequently facilitate a reverse intersystem crossing process from high-lying excited states and suppress possible energy loss pathways. On the strength of such a strategy, a series of new blue-emitting hot exciton materials based on non-aromatic pyrazoline derivatives was successfully fabricated and subsequently applied to OLED devices with EQEmax of 5.03%. These outcomes not only presented a series of new electroluminescent triplet-harvesting materials but also provided a practicable strategy for designing, screening and developing new generation luminogens for OLED applications.
https://pubs.rsc.org/en/content/articlelanding/2023/tc/d3tc01192e
