Formally Triplet Aluminyl Anions within the [Al2Pd2]2– Cluster Stabilized by All-Metal Double Aromaticity
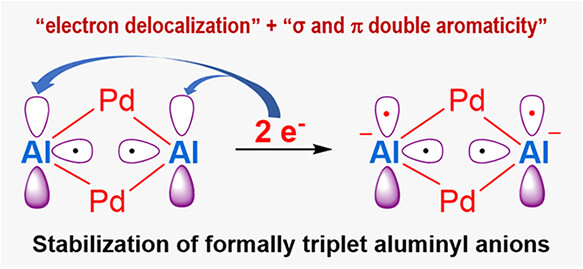
While triplet carbenes have been stabilized through spin delocalization, capturing triplet aluminyl anions─isoelectronic counterparts of carbenes─has remained an unsolved challenge in main-group chemistry. All previously reported aluminyl anions exhibit a singlet ground state, despite the use of diverse ligand frameworks. Herein, we report the first example of formally triplet aluminyl anions within bimetallic clusters, [((CH3)2C(CH2NPiPr2)2)2Al2Pd2]M2 (M = Li, Na, K). These clusters are obtained via chemical reduction of a σ-aromatic Al(II) precursor, [((CH3)2C(CH2NPiPr2)2)2Al2Pd2]. Combined structural and theoretical analyses reveal that each Al(I) center bears two electrons, which exhibit significant electron delocalization across the cluster. This delocalization confers four-center two-electron (4c-2e) σ and 4c-2e π double aromaticity to the [Al2Pd2]2– unit, providing the necessary stabilization for the two Al(I) centers. This work not only demonstrates the viability of formally triplet aluminyl anions but also establishes electron delocalization as a general strategy for stabilizing electron-rich main-group systems.
