A theoretical study on the mechanism of ruthenium(II)-catalyzed phosphoryl-directed ortho-selective C–H bond activations: the phosphoryl hydroxy group triggered Ru(II)/Ru(0) catalytic cycle
Submitted by Jun Zhu on Thu, 06/01/2017 - 10:01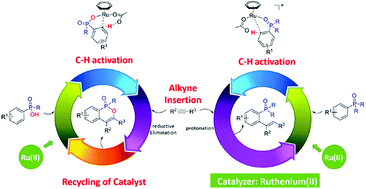
Using density functional theory (DFT) calculations, the present study explores the mechanisms of two ruthenium(II)-catalyzed phosphoryl-directed ortho-selective C–H bond activation reactions. Depending on the nature of the phosphoryl groups, namely R2P(O) versus RP(O)OH, two different products could be selectively synthesized. For R2P(O), the overall catalytic cycle includes three basic steps: C–H bond activation, alkyne insertion, and protonation. The oxidation state of the Ru center does not change during this catalytic process.