Nature Chemistry


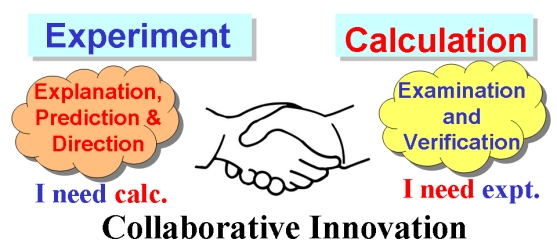

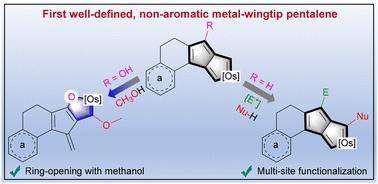
Since the concept of aromaticity was first introduced in transition metal complexes, metals have become a crucial component for modulating aromaticity, leading to a variety of structural frameworks. Initial studies successfully achieved aromatic pentalene dianions through metal-ion coordination, and recent advancements in bridgehead metallapentalenes have demonstrated the transformation of antiaromaticity into aromaticity.
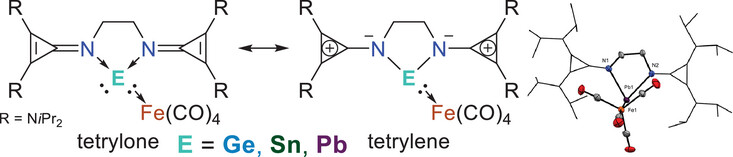
We report on the utilization of the ethylene-bridged bis[(dialkylamino)cyclopropenimine] (bisCPI) ligand, LCPI, to give access to new main-group E(II) halide complexes (E = Ge, Sn, Pb; 1, 2, 3). Subsequent reduction with Collman's reagent (Na2Fe(CO)4 • dioxane) enables the isolation of a series of zero-valent tetrylone-tetracarbonyl iron complexes, (LCPI)E(Fe(CO)4 (E = Ge (4), Sn (5), Pb (6)).
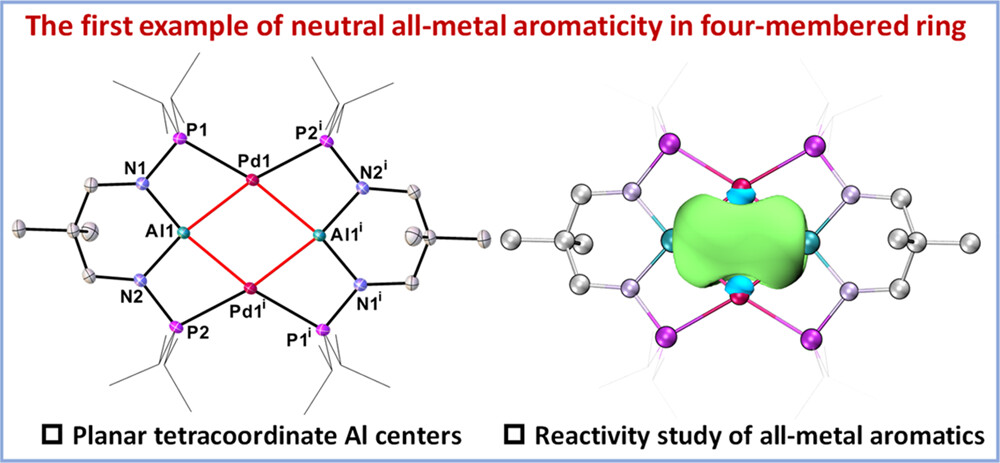
Aromaticity is a cornerstone concept in chemistry, playing a crucial role in understanding molecular stability and reactivity. Traditionally, aromaticity has been primarily associated with cyclic planar conjugated organic molecules composed solely of carbon, but it has recently expanded to include metal-containing systems. However, metal-only aromatics remain extremely scarce. Here, we present the first neutral all-metal aromatic cluster with a rhombic geometry.

Recent research has sparked significant interest in exploring the effects of BN unit doping on the electronic structure of isoelectronic and isostructural benzene analogs, driven by their promising applications in pharmaceuticals and material sciences. In this study, we provide the first comprehensive investigation of BN/CC isosterism in 2,5-dihydro-1,4,2,5-diazadiborinine and its isomers (1−13) through density functional theory (DFT) calculations and machine learning-based analysis.
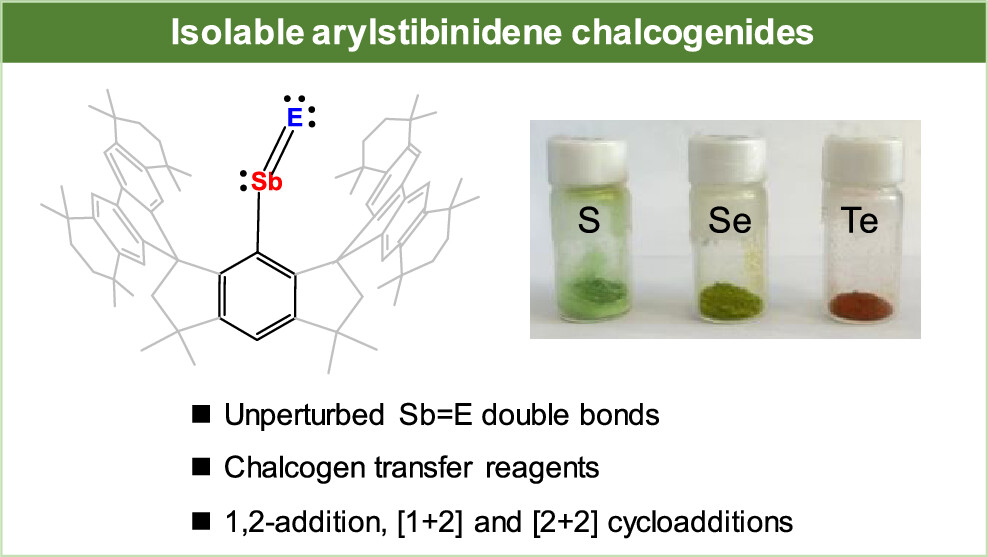
Nitroso compounds, R-N═O, containing N═O double bonds are ubiquitous and widely utilized in organic synthesis. In contrast, heavier congeners of nitroso compounds, namely pnictinidene chalcogenides R-Pn = E (Pn = P, As, Sb, Bi; E = O, S, Se, Te), are highly reactive and scarce. They have been stabilized in the coordination sphere of Lewis acid/base or by pronounced contribution from resonance structures, whereas free species with unperturbed pnictogen-chalcogen double bonds remains elusive.
The concept of adaptive aromaticity, which denotes two-state aromaticity in both the lowest singlet and triplet states, stands in marked contrast to the traditional one-state aromaticity governed by the Hückel and Baird rules. Nonetheless, organic compounds exhibiting adaptive aromaticity remain particularly rare.
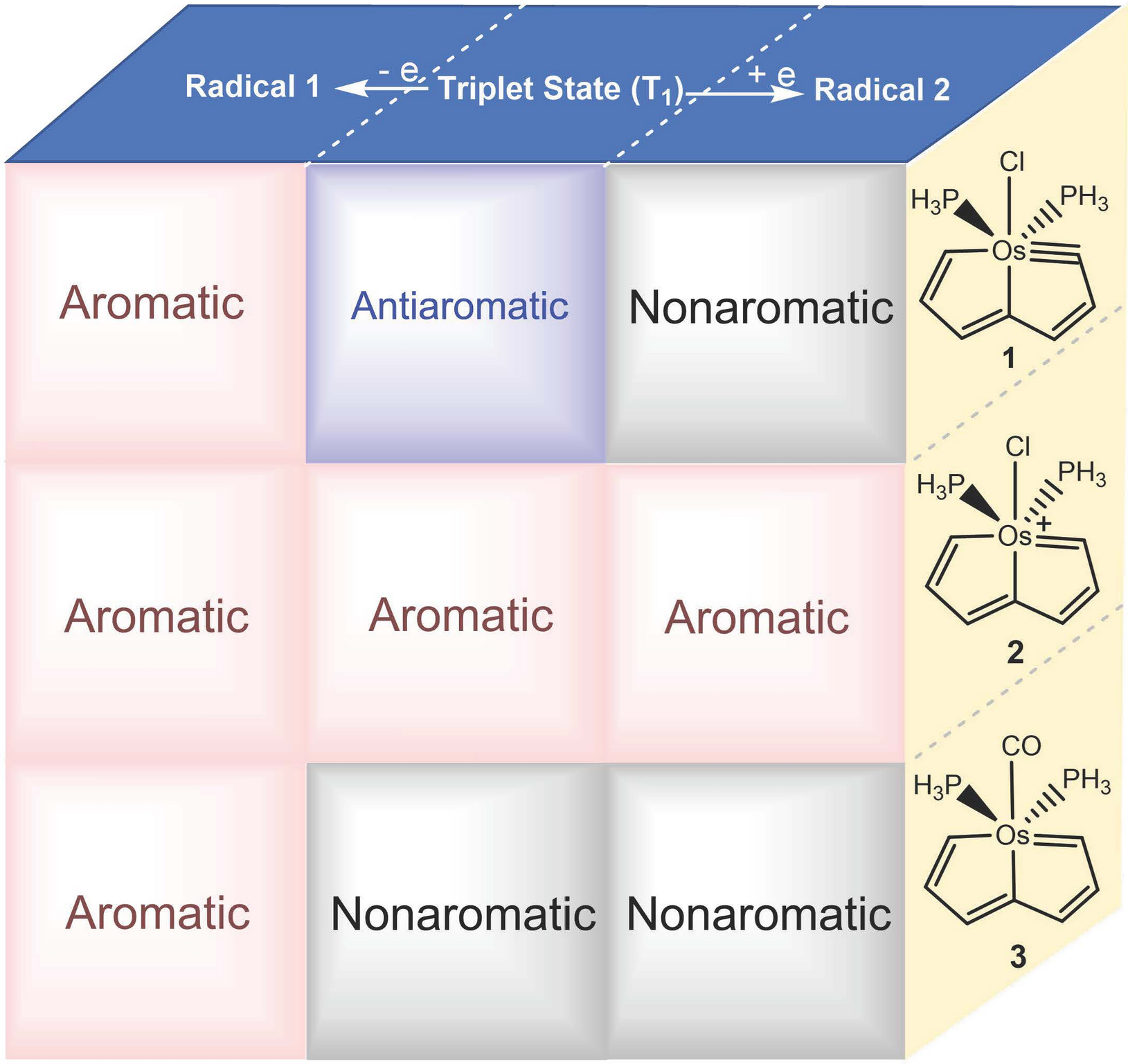
As one of the most important concepts in organic chemistry, aromaticity has attracted considerable attention from both theoretical and experimental chemists. Limited by the traditional rules (Hückel’s rules and Baird’s rules), species can only achieve aromaticity in a single state (S0 or T1) in most cases. In 2018, our group first reported 16 electron osmapentalene that showed aromaticity in both the S0 and T1 states, which is defined as adaptive aromaticity.
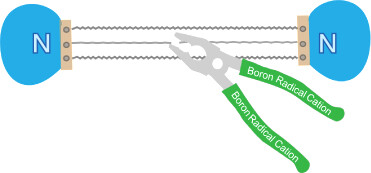
Activation of dinitrogen (N2) under mild conditions has been a particularly challenging project for decades, owing to the highly strong N≡N triple bond. In recent years, the main group species have emerged as a prominent strategy in the field of dinitrogen activation, but the reported examples remain particularly rare compared with transition metal complexes. Herein, we performed a comprehensive density functional theory (DFT) calculation of N2 activation by boron radical cations.
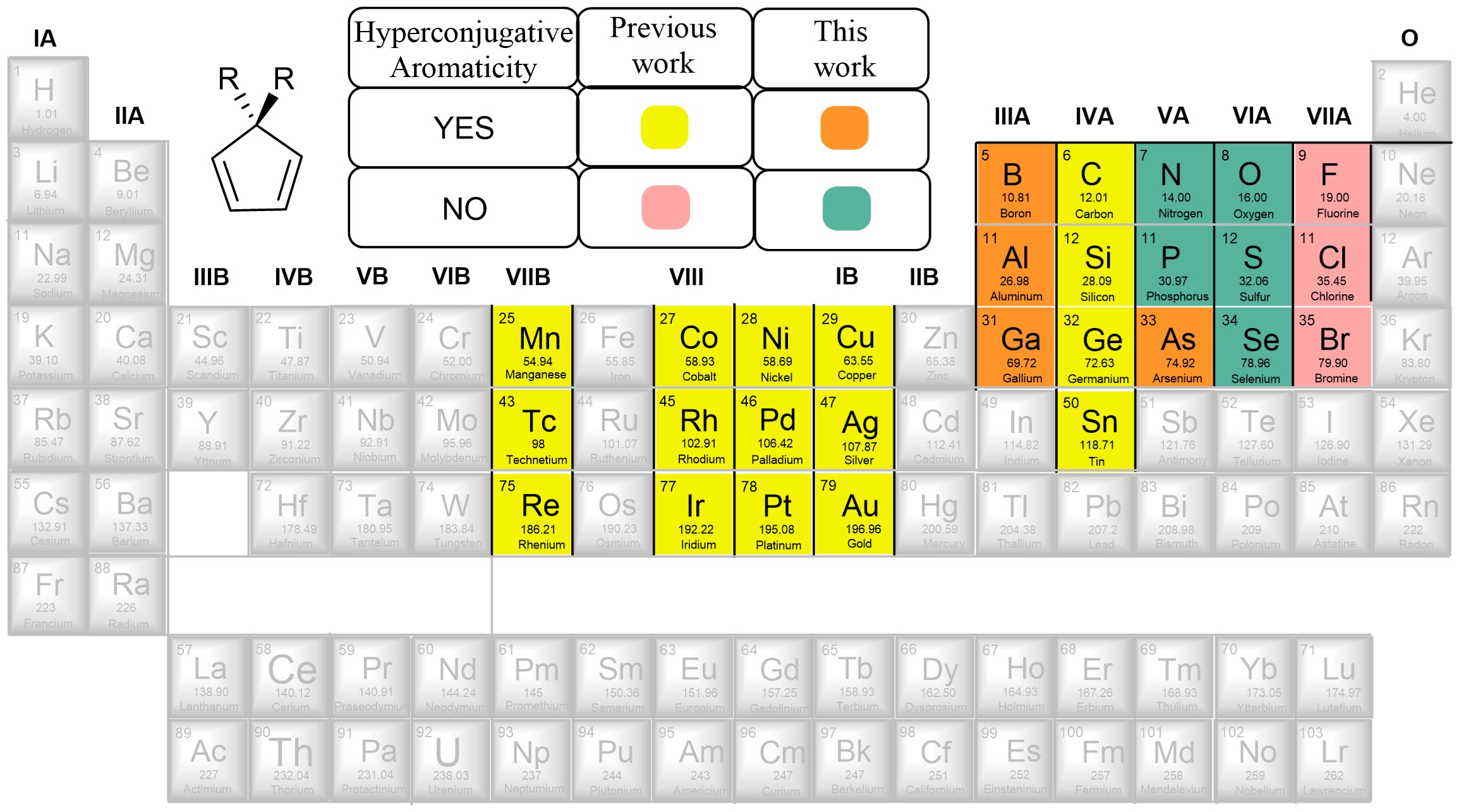
Combining aromaticity and hyperconjugation, two important concepts in organic chemistry, leads to hyperconjugative aromaticity, which was first proposed by Mulliken in 1939. However, previous studies on hyperconjugative aromaticity have mainly focused on substituents containing either main-group elements (group 14) or transition metals in groups 7, 9, 10, and 11. In this study, we perform density functional theory (DFT) calculations on cyclopentadiene and pyrrolium derivatives containing groups 13, 15 and 16 substituents to examine the possibility of achieving hyperconjugative aromaticity.
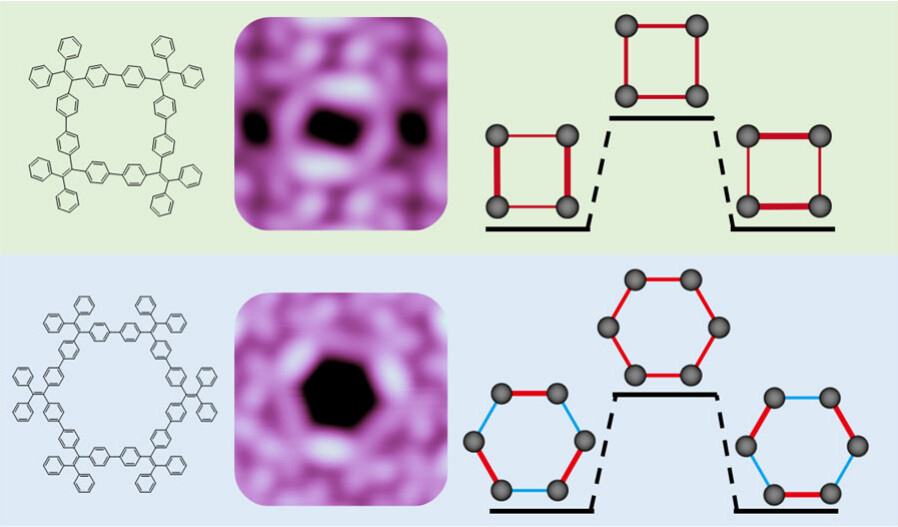
Tetraphenylethylene (TPE) is a prototype aggregate-induced emission molecule. TPE-based conjugated macrocycles exhibit unique optical properties due to their peculiar cyclic topology. Because the symmetry of macrocycles strongly affects their photophysical properties, here we report a single-molecule study of the structures and orbitals of two TPE-based macrocycles of (C26H18)4 and (C26H18)6. Using scanning tunneling microscopy and spectroscopy, we discover that both macrocycles undergo spontaneous symmetry breaking in their conformations and frontier orbitals.
Copyright © 2026,
Theme Originally Created by Devsaran
