Nature Chemistry



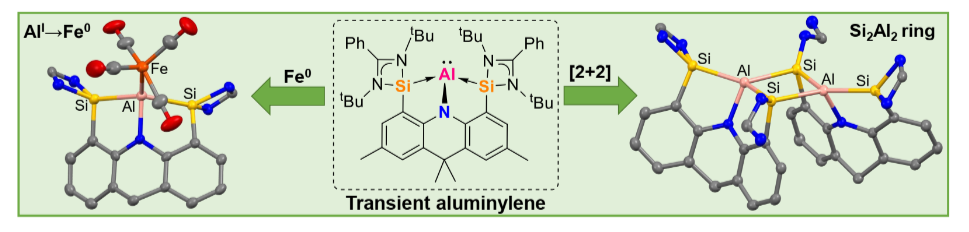
The suitability of electron-rich bis-silylenes, specifically the neutral chelating [SiII(Xant)SiII] ligand (SiII = PhC(NtBu)2Si, Xant = 9,9dimethylxanthene) and the anionic [SiII(NAcrid)SiII)]‒ pincer ligand (NAcrid = 2,7,9,9-tetramethylacridane), has been successfully probed to stabilize monovalent bis-silylene-supported aluminium complexes (aluminylenes).
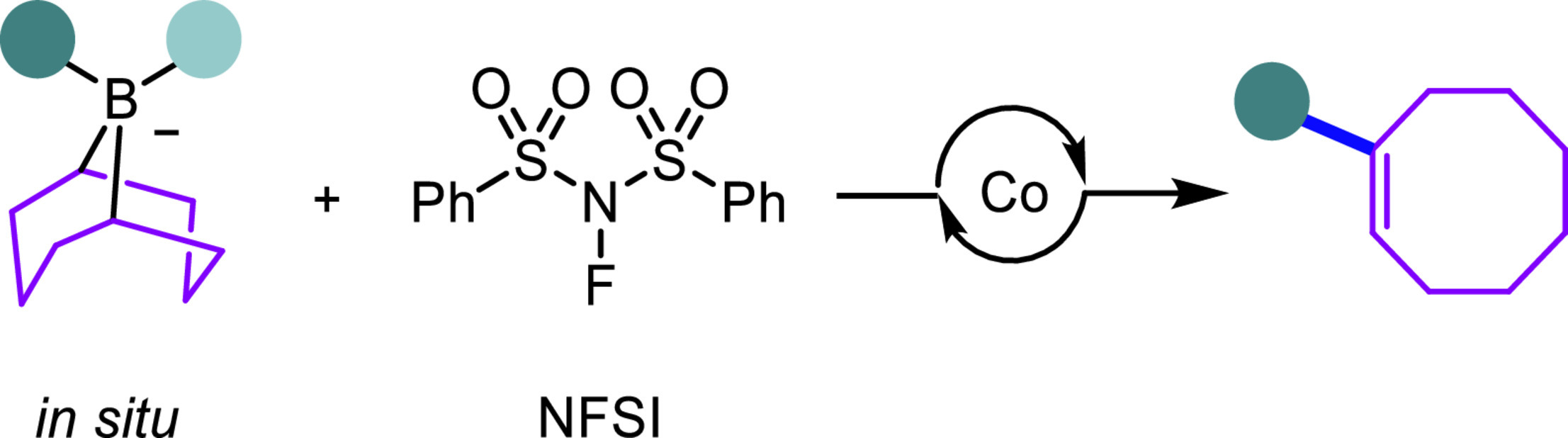
In most Suzuki-Miyaura carbon-carbon cross-coupling reactions, the borabicyclo[3.3.1]nonane scaffold (9-BBN) only serves as an auxiliary facilitating the transmetalation step and thus is transformed into by-products. There are rare examples where the 9-BBN derivatives serve as the potentially diverse C8 building blocks in cross-coupling reactions. Herein, we report a cobalt-catalyzed migratory carbon-carbon cross-coupling reaction of the in situ formed 9-BBN ate complexes to afford diverse aryl- and alkyl-functionalized cyclooctenes.
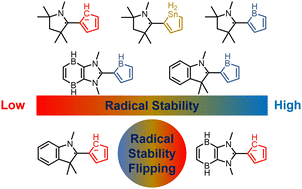
Aromaticity, as a classical and fundamental concept in chemistry, can enhance thermodynamic stability. In sharp contrast, a previous study showed that antiaromaticity rather than aromaticity can enhance the radical stability of α-methyl heterocyclic compounds. Here, we demonstrate a similar antiaromaticity-promoted radical stability when the methyl group is replaced by five-membered (alkyl)(amino)cyclics (AACs). More interestingly, when an AAC is fused with an antiaromatic ring, the radical stability could be either reduced or enhanced, depending on the spin population.
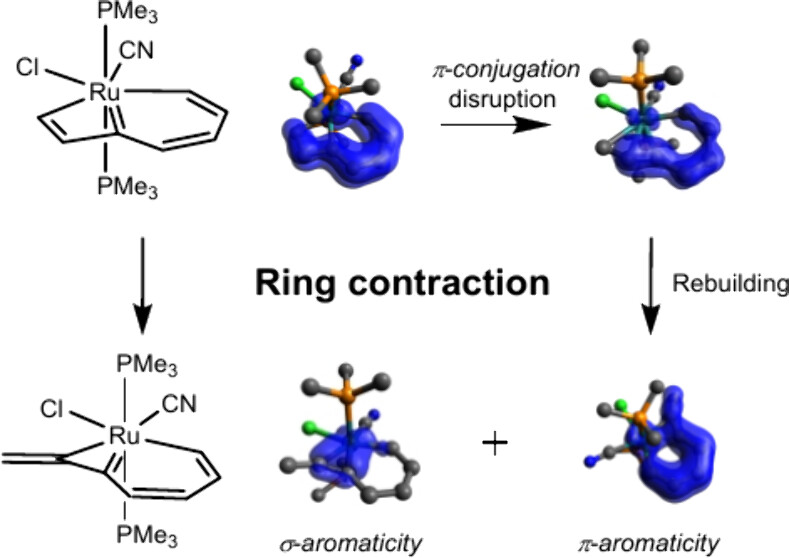
Ring contraction of metallacyclobutadiene to metallacyclopropene is rare because of the increasing strain from a four-membered ring to a three-membered one. Here we demonstrate a new series of reactions of metallabenzocyclobutadiene to metallabenzocyclopropene via density functional theory calculations. The results suggest that these reactions are thermodynamically favorable ranging from −17.4 to −29.4 kcal mol–1, and a low reaction barrier (10.3 kcal mol–1) is achieved when the metal center is Ru and the ligands are one cyanide and one chloride.
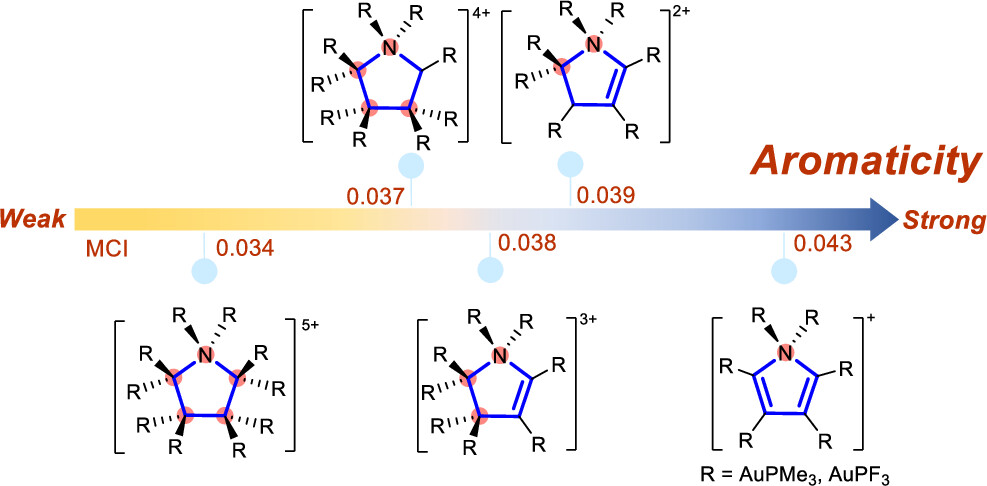
Aromaticity is a fundamental concept in organic chemistry. Hyperconjugative aromaticity, also known as hyperconjugation-induced aromaticity, has evolved from its origin from main group substituents to transition metal analogues, establishing itself as an important category of aromaticity. Additionally, aromatic compounds comprising two sp3-carbon atoms have recently been reported both experimentally and computationally. However, what is the maximum number of sp3-hybridized atoms needed to maintain hyperconjugative aromaticity?
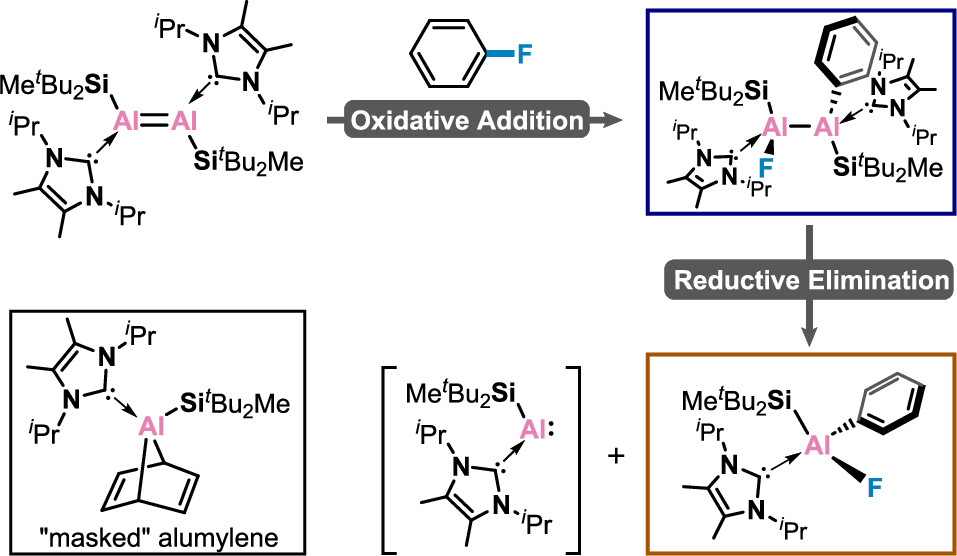
The activation of C–F bonds has long been regarded as the subject of research in organometallic chemistry, given their synthetic relevance and the fact that fluorine is the most abundant halogen in the Earth’s crust. However, C–F bond activation remains a largely unsolved challenge due to the high bond dissociation energies, which was historically dominated by transition metal complexes. Main group elements that can cleave unactivated monofluorobenzene are still quite rare and restricted to s-block complexes with a biphilic nature.
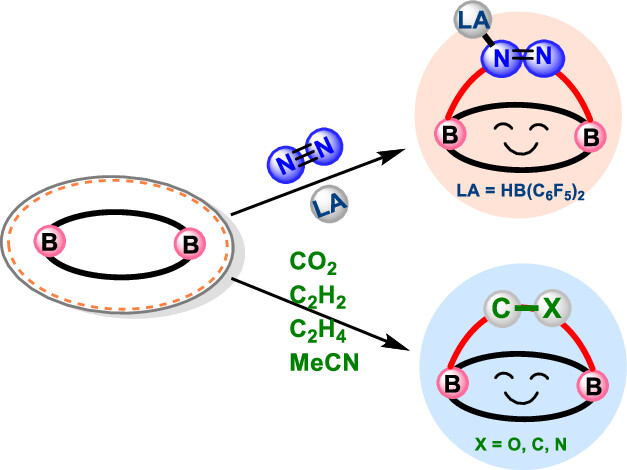
Although main group species have emerged in the field of dinitrogen activation in recent years, the reported examples are particularly rare in comparison with transition metal complexes due to their significant challenges. Herein, we demonstrate a [4 + 2] cycloaddition reaction of N2 (with an activation energy as low as 12.5 kcal mol–1) initiated by an inorganic benzene via density functional theory calculations. Such N2 activation is supported by the elongated nitrogen–nitrogen bond distance (dNN), decreased vibration frequency (νNN), and weakened Wiberg bond index (WBINN).
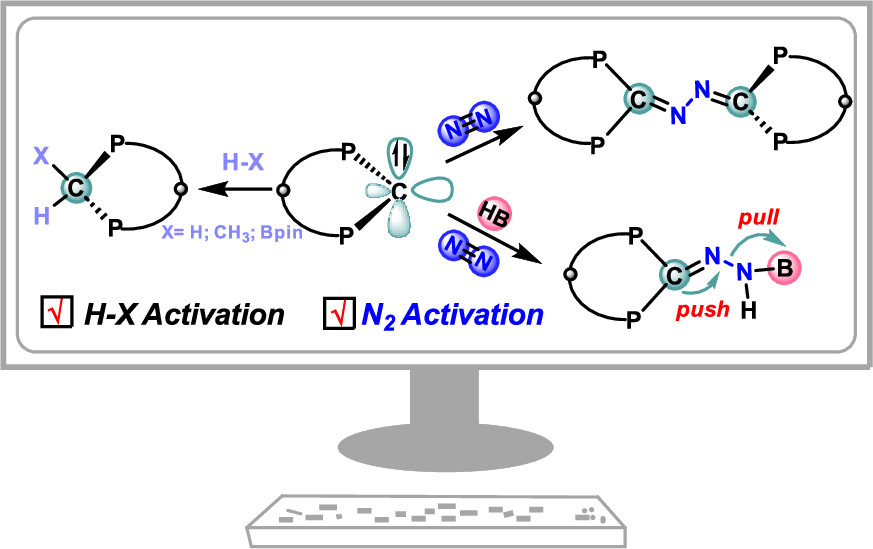
Although the main group species in the s and p blocks have begun to gain prominence in the field of dinitrogen (N2) activation in recent years, reports of carbene-mediated N2 activation remain particularly rare, especially for carbenes with a σ0π2 electronic configuration. Herein, we demonstrate examples of N2 activation initiated by a carbene with a σ0π2 electronic configuration and consequent N2 hydroboration reaction (with a reaction barrier as low as 19.9 kcal/mol) via density functional theory calculations.
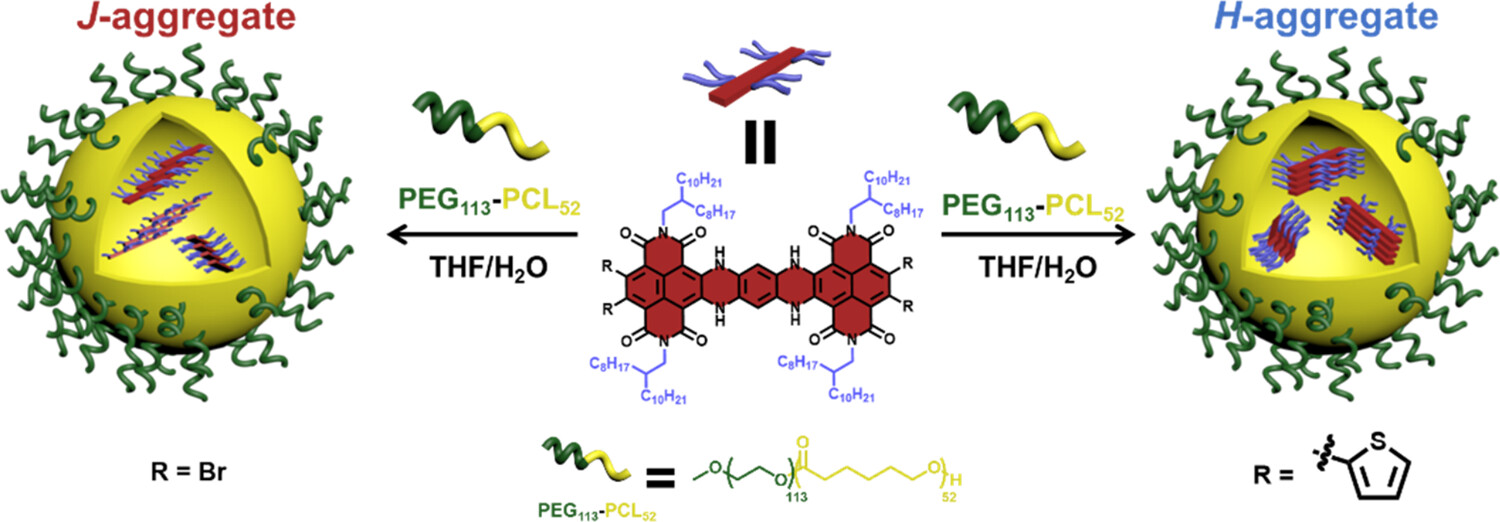
Rational control of the supramolecular aggregation of π-conjugated molecules plays an important role in determining their optoelectronic properties and applications. Here, we report a systematic study of the factors, including solvent polarity, concentration, and surfactants, that affect the aggregation behavior of a brominated hydroazaheptacene tetraimide (HATI) and its thiophene-substituted derivative, Th-HATI, as near-infrared fluorophores, in both nonpolar and polar solvents.
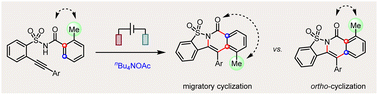
Efficient control over several possible reaction pathways of free radicals is the chemical basis of their highly selective transformations. Among various competing reaction pathways, sulfonimidyl radicals generated from the electrolysis of 2-alkynylbenzenesulfonamides undergo cascade migratory or ortho-cyclization cyclization selectively.
Copyright © 2026,
Theme Originally Created by Devsaran
