Nature Chemistry




Aromaticity generally describes a cyclic structure composed of sp2-hybridized carbon or hetero atoms with remarkable stability and unique reactivity. The doping of even one sp3-hybridized atom often damages the aromaticity due to the interrupted electron conjugation. Here we demonstrate the occurrence of an extended hyperconjugative aromaticity (EHA) in a metalated indole ring which contains two gem-diaurated tetrahedral carbon atoms. The EHA-involved penta-aurated indolium shows extended electron conjugation because of dual hyperconjugation.

Benzene, the prototype of aromatics, has six equivalent C‐C bonds (1.397 Å), which are intermediate between a C‐C double bond and a C‐C single bond. For over 80 years, chemists have spent much effort on freezing a localized structure to obtain a distorted bond‐length alternating benzene ring in the ground state, leading to various localized trisannelated benzene rings. However, most of the central benzene rings are still aromatic or nonaromatic. Here we report an antiaromatic benzene ring caused by hyperconjugation.
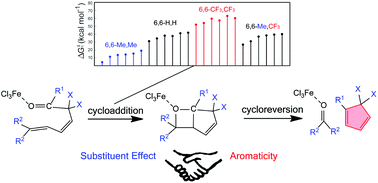
Olefin metathesis is a fundamental organic reaction of great importance that led to the 2005 Nobel Prize in Chemistry. As a variation of olefin–olefin metathesis, carbonyl–olefin metathesis (COM) is less developed, but still significant progress has been made recently. However, how the aromaticity affects the reaction mechanisms remains unclear. Here we perform density functional theory calculations on iron(III) catalyzed COM in 2,5- and 3,5-hexadienals.

Pincer complexes are a remarkably versatile family benefited from their stability, diversity, and tunability. Many of them contain aromatic organic rings at the periphery, and aromaticity plays an important role in their stability and properties, whereas their metallacyclic cores are not aromatic. Herein, we report rhodapentalenes, which can be viewed as pincer complexes in which the metallacyclic cores exhibit considerable aromatic character. Rhodapentalenes show good thermal stability, although the rhodium-carbon bonds in such compounds are fragile.
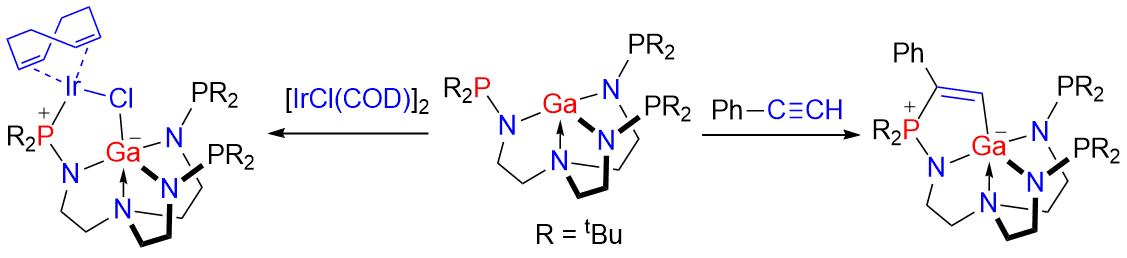
Frustrated Lewis pairs (FLPs) represent a new paradigm of main‐group chemistry. The Lewis acidic centers in FLP chemistry are typically B and Al atoms in the studies reported over the past decade, and most of them are tri‐coordinated with strong electron‐withdrawing groups. Herein, we report a Ga/P system containing an unprecedented four‐coordinated Lewis acidic Ga center. This Ga/P species performs classical addition reactions toward heterocumulenes, alkyne, diazomethane, and transition metal complex. Regioselective formation of the products can be rationalized by DFT calculations.
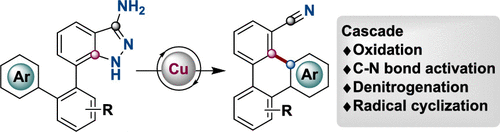
We present a novel Cu-catalyzed aromatic metamorphosis of 3-aminoindazoles via oxidative cleavage of two C–N bonds of 3-aminoindazoles. This unprecedented reactivity of 3-aminoindazoles allows one to forge diverse nitrile-containing triphenylenes in decent yields via generation of the cyano group in situ. The current study reveals that 3-aminoindazoles could be harnessed as radical precursors via oxidative denitrogenation, the reaction mechanism of which was supported by density functional theory calculations.

Unusual 1,2‐migration reactions of N‐heterocyclic carbene (NHC) on transition metals were investigated using density functional theory calculations. Our results reveal that the electronic properties, ring strain of the four‐membered ring, and aromaticity of NHC play crucial roles in the thermodynamics of such a 1,2‐migration.
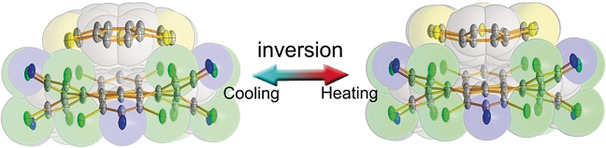
Bowl inversion is a unique property of buckybowls. The polarity and assembly configuration of buckybowls are reversed after bowl inversion. So far, this unique phenomenon has been studied in solution and on surface, but not in solid state due to spatial constraint. Now a series of exo‐type supramolecular assemblies of trithiasumanene and nanographene are investigated. Tuning the electron density of the nanogaphene component was found to directly affect the binding constant of the complex.
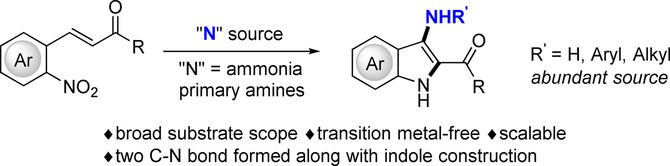
A step‐economic strategy for 3‐aminoindoles synthesis with ammonia or primary amines as “N” source under transition‐metal‐free conditions was achieved. A series of 3‐aminoindoles was obtained with abundant “N” source featuring high efficiency, mild conditions, environmental friendliness and scalability. Efficient syntheses of the intermediates of COX‐2 inhibitor and tubulin polymerization inhibitor were successfully accomplished with this newly developed strategy.

The [1,5]‐migration reaction has attracted considerable attention from experimentalists and theoreticians for decades. Although it has been extensively investigated in various systems, studies on pyrrolium derivatives are underdeveloped. Herein, a theoretical study on the reaction mechanism of [1,5]‐migration in both pyrrolium and pyrrole derivatives is presented.
Copyright © 2026,
Theme Originally Created by Devsaran
